ResusNation #157

Welcome to ResusNation #157
Thank you to everyone who completed the survey. Exciting changes coming soon!

The Science of Extreme Friendliness
Evolution looked at the wolf—a calculated apex predator with a healthy sense of boundaries—and deleted a handful of genes to see what would happen. What it produced was the dog. We used to think domestication was a sophisticated, multi-generational contract of "I give you scraps, you don't eat my kids," but it may actually be closer to a genomic glitch. Researchers have found that the extreme sociability separating your Golden Retriever from a forest-dwelling murder puppy is linked to structural variants in GTF2I and GTF2IRD1—the same genes implicated in the Williams-Beuren Syndrome critical region in humans. Where wolves remain reserved, wary, and socially selective, dogs are genetically wired toward indiscriminate friendliness with humans, including complete strangers.
The parallel in humans is striking. Williams Syndrome (WS), typically caused by a microdeletion on chromosome 7, produces a characteristic profile of hypersociability, heightened empathy, "elfin" facial features, cardiovascular anomalies, and mild-to-moderate intellectual disability. Individuals with WS show a near-complete absence of social fear—they approach strangers readily and form immediate emotional connections, often at the cost of appropriate social caution. Dogs show a remarkably similar behavioral signature: reduced stranger-avoidance, prolonged gaze toward human faces, and a strong drive for social contact that persists even when no food reward is offered. Both populations also show parallels in cognitive profile, with relative weaknesses in spatial reasoning and problem-solving alongside preserved or heightened social cognition. The comparison suggests that what we call "Man's Best Friend" behavior isn't simply a product of training or shared history—it's a hypersociability phenotype that appears to have been positively selected during domestication, with the same genomic region driving convergent social behavior across two very different species.

Enjoy ResusX, From Anywhere
Can't make it to Philly? No problem.
ResusX:2026 goes live May 18–20 — and you don't need a plane ticket to be in the room. The Virtual Package puts every session on your screen, in real time, the moment it happens.
Same speakers. Same debates. Same procedural demos. You're just watching from a better chair.
Here's what you get:
→ Full livestream of every session — all three days, as it happens. Not highlights. Not a recap blog. The whole thing.
→ On-demand replays so you can pause, rewind, and rewatch the parts that rewire your thinking. At 2am. On your next shift break. Whenever it clicks.
→ Live Q&A — submit questions directly to faculty during sessions.
→ CME and CEU credits — same credits as the in-person attendees. No difference.
→ Expert debates, live procedural demos, real cases, audience polling — the full ResusX experience, streamed to wherever you are.
We're talking resuscitative thoracotomy. Impella placement. Perimortem C-section. Bougie-assisted cric. Proning demos. POCUS for dissection. Three days of the sharpest resuscitation education anywhere — and you won't miss a second of it.
You Got 2 Choices:
→ Conference Pass — $247 (40% off): Livestream access to every session.
→ All-Access Pass — $479 (40% off): Livestream + on-demand replays so you can revisit everything after the conference ends.
Look — nothing replaces being in that comedy club where the energy is dialed to eleven. We know that. But this is the next closest thing: every minute of content, on your terms, with a replay button.
If you've been telling yourself you'll level up your resuscitation skills "soon" — this is what soon looks like. No flights. No hotel. No asking someone to cover your shifts for three days.
Can 1-Axis Completely Change Your IVC Assessment?
When it comes to assessing the IVC for fluid responsiveness, most clinicians are doing it wrong — and I say that having watched it taught incorrectly for years. The standard approach is the long axis view, and I get why it feels intuitive. But here's the problem: to get an accurate measurement, you need to be perfectly centered on that vessel. Even a slight off-axis view and you're already underestimating the size and overestimating collapsibility. That's not a minor technical footnote — that's an error that can directly influence your fluid management decisions.
The fix is simpler than you think: use the short axis. It gives you a far more reliable and reproducible assessment of both IVC size and collapsibility without the same margin for error that plagues the long axis approach. This is one of those small technical adjustments that separates good point-of-care ultrasound from great point-of-care ultrasound. If you're making clinical decisions about fluid tolerance based on IVC assessment, you owe it to your patients to be using the most accurate method available — and right now, that's the short axis view.
Watch the full video here and leave a comment.
Don't forget to like and follow my IG, TikTok, YT, Facebook or LinkedIn accounts.

The debate over RSI has moved beyond traditional dogmas, focusing on the specific physiological profiles of induction agents and paralytics. While etomidate remains a reliable drug for its hemodynamic neutrality, its association with adrenal suppression and its pro-convulsant nature make ketamine a superior choice for septic or hypotensive patients. Conversely, propofol is increasingly criticized for RSI due to its tendency to cause significant hypotension and its "quick on, quick off" duration, which risks leaving a patient unsedated during the critical moments of laryngoscopy. For patients requiring a more prolonged sympatholytic effect, such as those in alcohol withdrawal or stimulant toxicity, high-dose midazolam is presented as a viable, albeit slower-acting, alternative.
Regarding paralytics, the consensus heavily favors rocuronium over succinylcholine. The historical argument for succinylcholine—that its short duration provides a safety net in "can't intubate, can't ventilate" scenarios—is largely dismissed as a myth, as the drug rarely wears off fast enough to prevent desaturation in a critically ill patient. Rocuronium, when dosed correctly at 1.2 mg/kg, provides identical intubating conditions and a longer duration of action that ensures the patient remains paralyzed until the airway is secured. Furthermore, the availability of reversal agents like sugammadex has effectively neutralized the concerns regarding neurological exams, making rocuronium the safer, more dependable standard for modern airway management.
Check out this video of Dr. Reuben Strayer from ResusX:2025 now!

 Is HFNO Actually Saving Lives in Acute Respiratory Failure?
Is HFNO Actually Saving Lives in Acute Respiratory Failure?
Acute hypoxemic respiratory failure is the single biggest driver of ICU admissions worldwide, and for the past decade high-flow nasal cannula (HFNC) oxygen has been the go-to first-line intervention. The SOHO trial set out to test whether HFNC actually reduces mortality. This multicenter, open-label, randomized trial enrolled 1,110 adult ICU patients across 42 French centers with severe acute hypoxemic respiratory failure — defined by a PaO₂:FiO₂ ratio ≤200, respiratory rate >25 breaths/minute, and bilateral pulmonary infiltrates. Patients were randomized to receive either HFNC at ≥50 L/min or standard oxygen via a non-rebreather mask, with a primary endpoint of 28-day mortality. The headline result was striking in its symmetry: 28-day mortality was identical at 14.6% in both groups (81 deaths each, P=0.98). HFNC did reduce intubation rates (42.4% vs. 48.4%) and improved dyspnea scores, respiratory rates, and subjective breathlessness within the first hour — but these physiological wins didn't translate into survival benefit, and the intubation reduction only barely crossed statistical significance.
The bottom line for practice: HFNC remains a reasonable first-line choice for acute hypoxemic respiratory failure, particularly where patient comfort and intubation avoidance are priorities — but clinicians should abandon the assumption that it reduces death compared to a well-managed non-rebreather mask. Resource-limited settings may reasonably continue using standard oxygen without compromising survival, and the modestly higher serious adverse event rate with HFNC (pneumothorax: 1.8% vs. 0.7%) should factor into individualized decisions alongside patient comfort and local resources.
My Takeaway Points:
-
Finding - 28-day mortality was identical between high-flow and standard oxygen groups (14.6% each; P=0.98), with a borderline reduction in intubation rates (42.4% vs. 48.4%, absolute difference −5.93 percentage points)
-
Practice Impact - HFNC should no longer be selected primarily for its presumed mortality benefit over standard oxygen — clinical decision-making should shift toward patient comfort, intubation risk, and resource availability
-
Population - Adult ICU patients with severe acute hypoxemic respiratory failure (PaO₂:FiO₂ ≤200, RR >25), predominantly viral or bacterial pneumonia (88%), including COVID-19; 22% immunocompromised; conducted across 42 French ICUs from 2021–2024
-
Limitation - The trial was powered to detect a 6 percentage point mortality difference, but actual mortality (14.6%) was lower than the 18% assumed for standard oxygen — reducing statistical power to detect smaller real differences; high glucocorticoid use (62%) may have lowered baseline mortality in both arms
Want to learn more? Read the full trial High-Flow or Standard Oxygen in Acute Hypoxemic Respiratory Failure (SOHO) by J-P. Frat, et al. in the New England Journal of Medicine.

Syncope: A Practical Approach That Won’t Miss the Killer Diagnoses
This week, Dr. Jain describes his structured approach to ED syncope, framed around three questions: is this actually syncope?, is there a lethal cause?, and can the patient safely go home? He stresses first confirming true syncope (transient loss of consciousness from global cerebral hypoperfusion with spontaneous full recovery) and ruling out mimics like seizure, hypoglycemia, and psychogenic pseudosyncope. Stabilization comes before history — ABCs, monitor, ECG, glucose, and IV access — with immediate resuscitation for red flags like hypotension, chest pain, hypoxia, focal neuro deficits, or signs of GI bleed. The core workup rests on four pillars: a focused history (circumstances, prodrome, cardiac background, medications, family history of sudden death), examination including orthostatics and cardiac/neuro exams, a mandatory ECG pattern-scanned for sudden-death signatures using the WOBBLER mnemonic (WPW, Obstruction/strain, Brugada, Blocks/brady, Long/short QT, Epsilon/ARVC, Regional ischemia), and targeted testing rather than shotgun panels. The must-not-miss list is summarized as FAST + B: Fatal arrhythmia, Aortic catastrophe, Structural heart disease, Thromboembolism, and Bleeding.
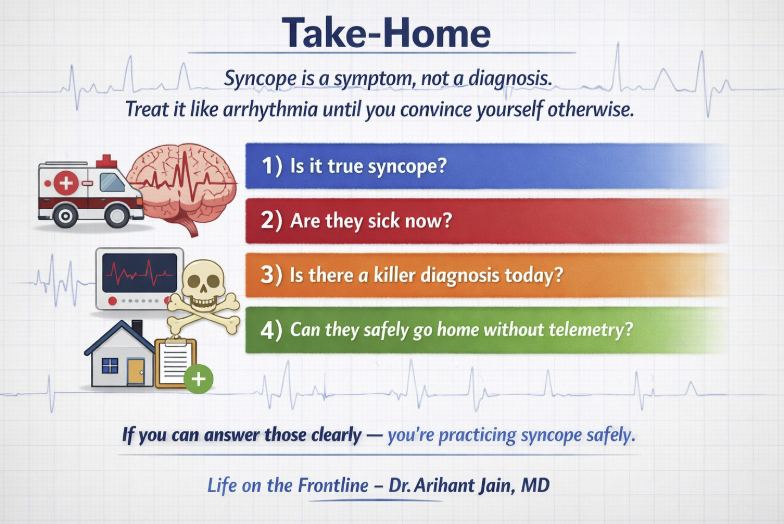
On risk stratification, Dr. Jain argues scores are not for every patient — benign vasovagal stories need no score and can be discharged on clinical grounds, while clear high-risk red flags mandate admission regardless of any score. Risk tools belong only in the indeterminate middle group after initial ED evaluation. Among the options, the Canadian Syncope Risk Score (CSRS) is favored for its strong discrimination (external-validation AUC around 0.91) and incorporation of physician gestalt; the San Francisco Syncope Rule is simple but overly nonspecific and tends to cause over-admission; and the Risk Stratification of Syncope in the Emergency Department (ROSE) Rule offers better specificity but depends on labs like BNP, making it less practical. Dr. Jain recommends a detailed discharge checklist: hemodynamically stable, a plausible benign mechanism (reflex/vasovagal, situational, or orthostatic), no high-risk historical features, a WOBBLER-negative ECG, no suspicion for dangerous alternative diagnoses (GI bleed, PE, dissection, ectopic, SAH), and a safe home situation with clear follow-up. For grey-zone cases, CSRS guides the call between discharge with expedited follow-up, observation, or telemetry admission.
Read the full post here and review this week's Frontline clinical pearls on IG.
----------
Dr. Arihant Jain is an Emergency Medicine physician at All India Institute of Medical Sciences (AIIMS) in New Delhi, and the creator of Life on the Frontline, a blog sharing concise, evidence-based insights from the ED. He currently serves as a Decision Editor for CPC-EM, is among the youngest ATLS faculty in India, and an AHA-certified BLS/ALCS instructor.
Connect with Dr. Jain: @humans.of.em (IG)
Watch the April Videos Now!

If you're an All-Access member, you're in for some great content this month. We have FIVE videos hand-picked by our staff that are high-yield and our most highly watched. We're featuring:
- Rezaie on "How High Can You Go?"
- Repanshek on "Crashing Anaphylaxis"
- Wright on "The Rapid Neuro Exam"
- Quazi on "Resuscitative Hysterotomy"
- Swaminathan on "Fast AF (How Do You Manage Atrial Fibrillation)"
Each month we bring you fresh new content from the best of the best in resuscitation. If you're an All-Access member, go watch these videos NOW!





Responses